An Open Label Clinical Trial to Evaluate the Efficacy and Tolerance of a Retinol and Vitamin C Facial Regimen in Women With Mild-to-Moderate Hyperpigmentation and Photodamaged Facial Skin - JDDonline - Journal
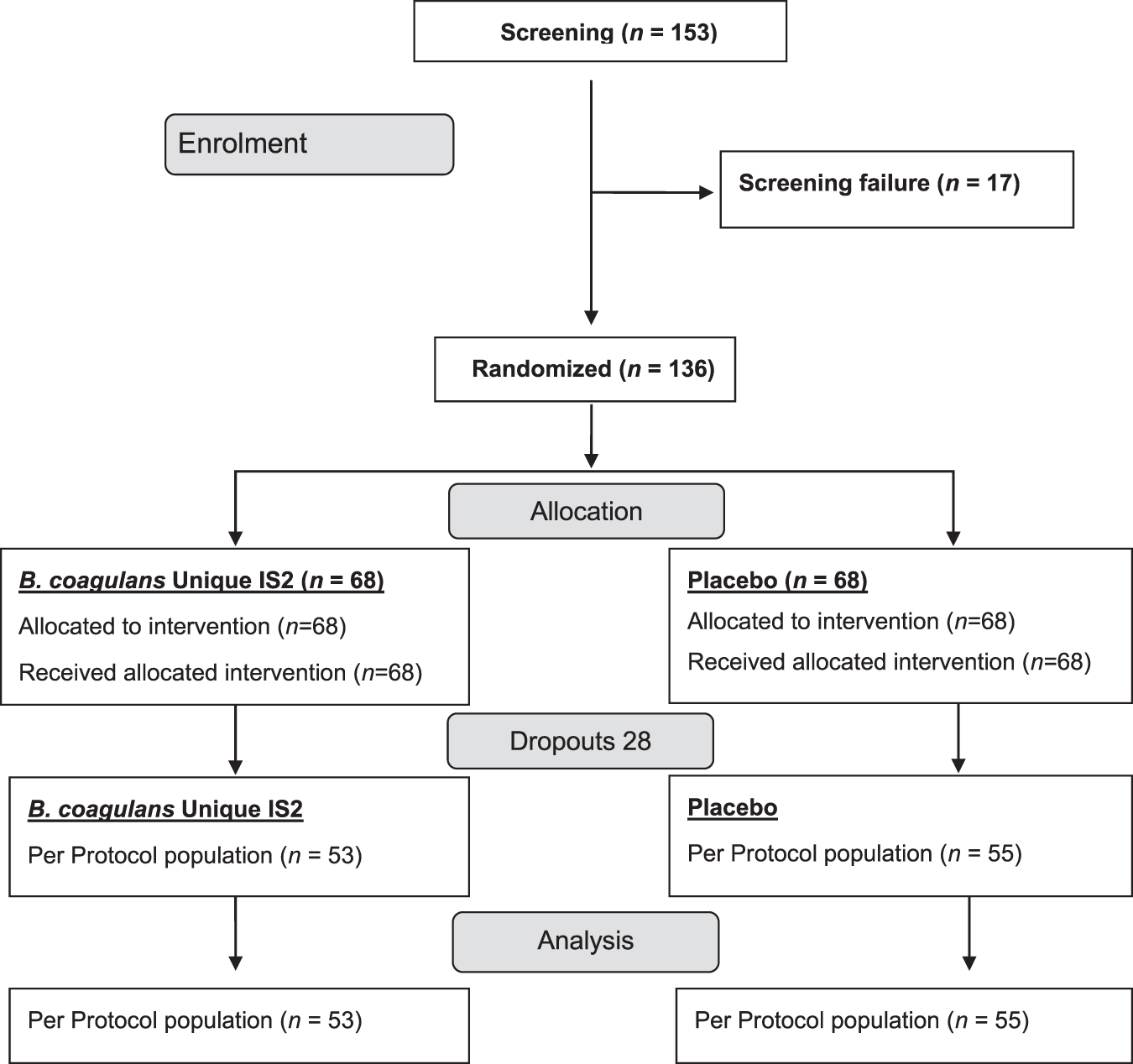
Randomized clinical trial: the effect of probiotic Bacillus coagulans Unique IS2 vs. placebo on the symptoms management of irritable bowel syndrome in adults | Scientific Reports
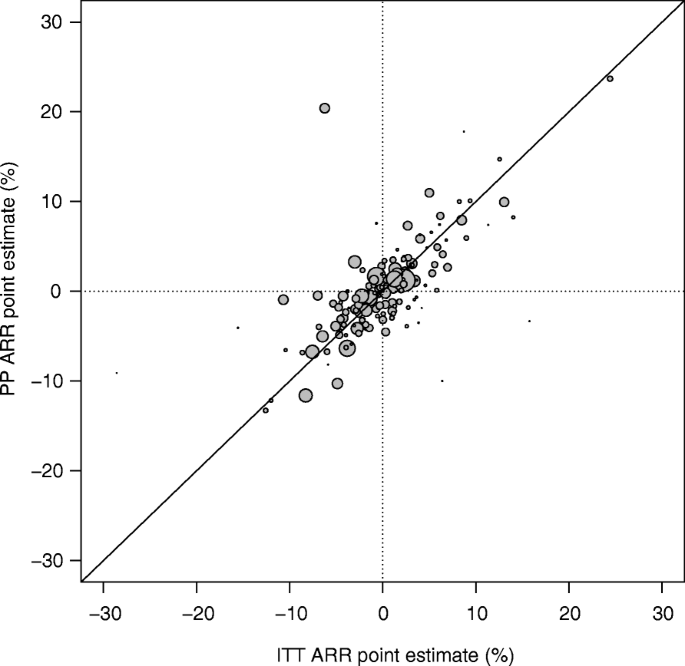
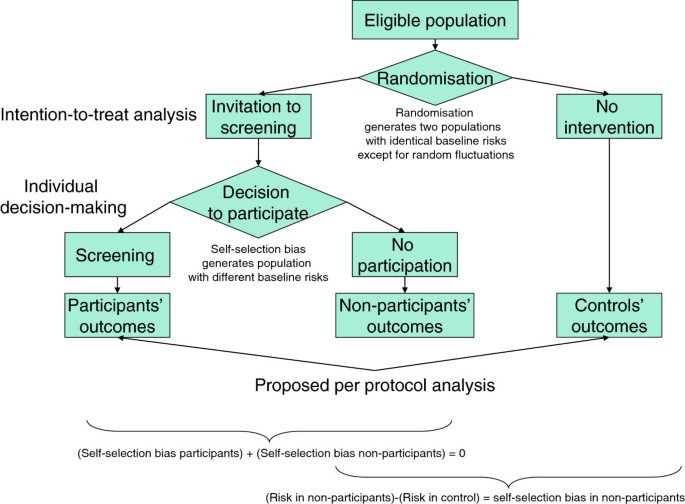
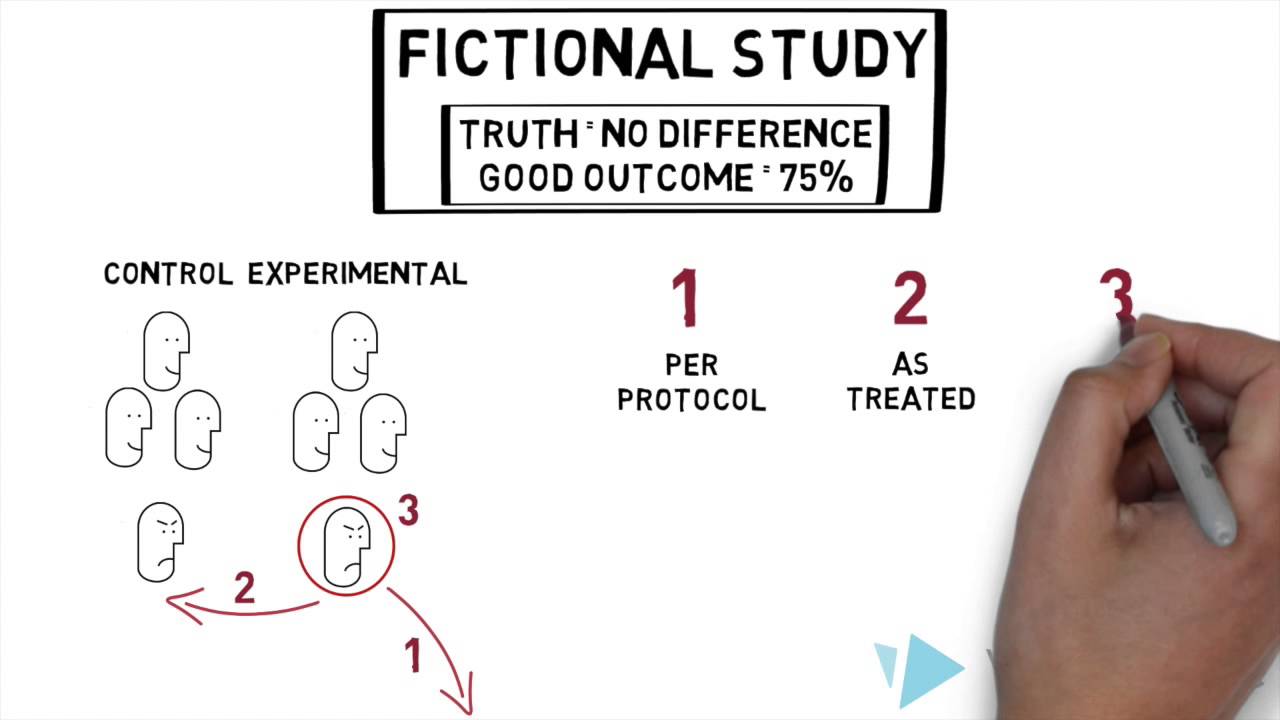
Intention-to-treat analysis may be more conservative than per protocol analysis in antibiotic non-inferiority trials: a systematic review | BMC Medical Research Methodology | Full Text

New regimens of benznidazole monotherapy and in combination with fosravuconazole for treatment of Chagas disease (BENDITA): a phase 2, double-blind, randomised trial - The Lancet Infectious Diseases

Figure 2, Forest Plot For ACR20 Responses at Week 24 in SUMMACTA and MUSASHI Studies for the Per-Protocol Population - Tocilizumab (Actemra) - NCBI Bookshelf

Trial profile. Population: ITT = intention-to-treat, PP = per protocol. | Download Scientific Diagram
Efficacy and Safety of the RTS,S/AS01 Malaria Vaccine during 18 Months after Vaccination: A Phase 3 Randomized, Controlled Trial in Children and Young Infants at 11 African Sites | PLOS Medicine

Denosumab biosimilar in postmenopausal osteoporotic women: A randomized, assessor-blind, active-controlled clinical trial